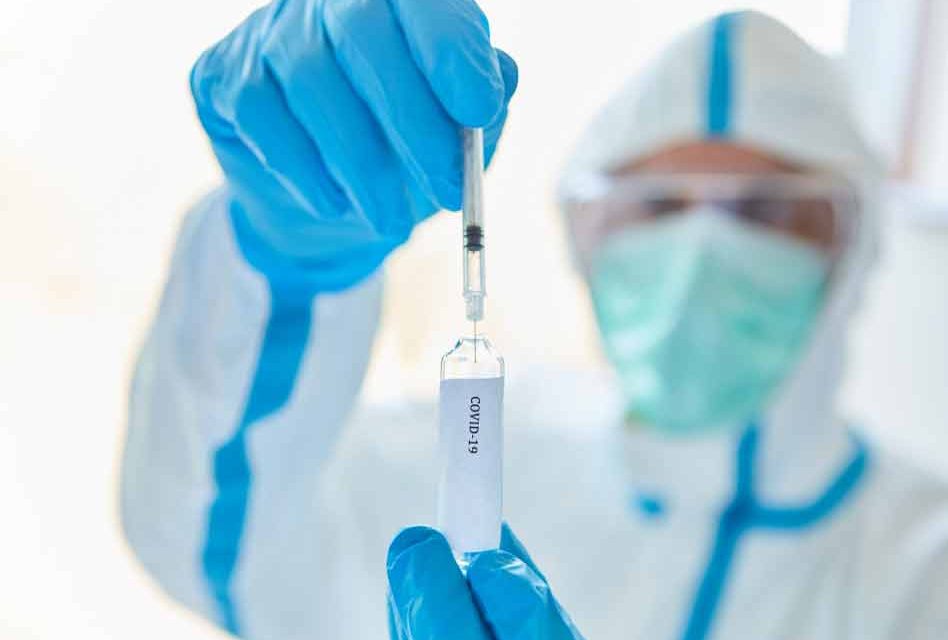
The U.S. Food and Drug Administration has issued emergency use authorization (EUA) for a vaccine for the prevention of COVID-19 in individuals 16 years of age and older. The emergency use authorization allows the Pfizer-BioNTech COVID-19 Vaccine to be distributed in the U.S. and is the first emergency use authorization ever issued in the United States.
“The FDA’s authorization for emergency use of the first COVID-19 vaccine is a significant milestone in battling this devastating pandemic that has affected so many families in the United States and around the world,” said FDA Commissioner Stephen M. Hahn, M.D.
Currently, more than 294,000 people have died from COVID-19 in the United States and states across the nation continue to see surging cases despite continued efforts to socially distance and wear masks.
Approval of the Pfizer vaccine marks a major milestone in the United States’ fight against the COVID-19 pandemic. Distribution of the vaccine is expected to begin almost immediately and take months to complete. However, according to health officials, phase 3 clinical trials did not show whether the vaccine will prevent asymptomatic infection or prevent people from transmitting the virus to others.
So, it’s likely that mask-wearing and physical distancing will continue to be a part of our lives in the United States well into next year to help slow the spread of the virus.
Many health experts emphasize that vaccines don’t save lives — vaccination does.
By the end of this month, 6.4 million doses of the Pfizer/BioNTech vaccine will be available in the United States, according to federal officials.
About half of those doses will begin shipping to states within 24 hours of the FDA’s emergency approval. The rest will be distributed later when the initial recipients are due to receive their second dose about 3 weeks later.
Preparation for distribution began months ago with coordination between federal and local health officials, as well as with healthcare systems and pharmacy chains.
FedEx and United Parcel Service, are the two companies expected to handle the bulk of vaccine deliveries.